Table of Contents
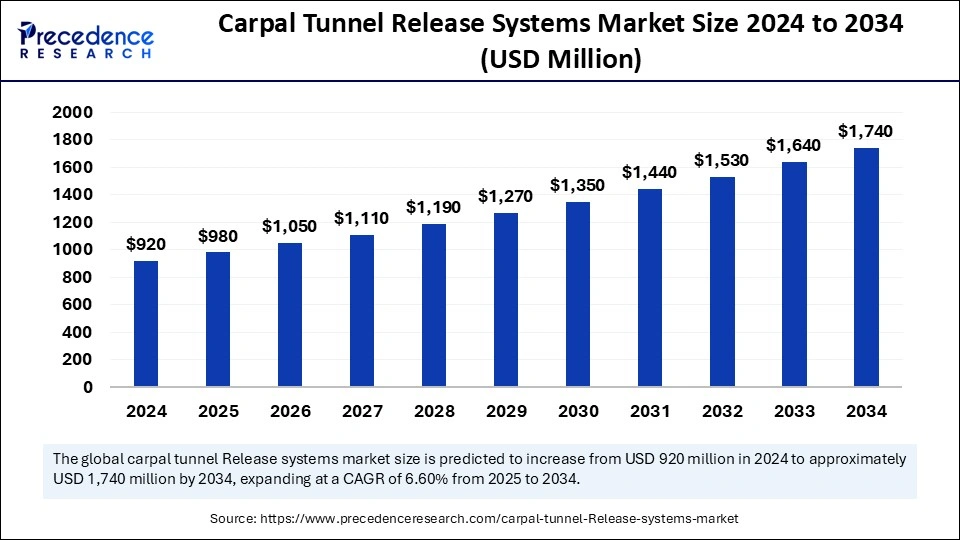
The global carpal tunnel release systems market size was valued at USD 920 million in 2024 and is expected to reach around USD 1,740 million by 2034, CAGR of 6.60%.
Get a Free Sample Copy of the Report@ https://www.precedenceresearch.com/sample/5763
Carpal Tunnel Release Systems Market Key Highlights
-
North America led the market in 2024, capturing a dominant 49% share.
-
Asia Pacific is forecasted to experience the most rapid market expansion in the coming years.
-
The open carpal tunnel release systems segment held a substantial 59% market share in 2024.
-
The endoscopic carpal tunnel release systems segment is projected to grow at the highest CAGR from 2025 to 2034.
-
Hospitals accounted for a significant portion of the market in 2024.
-
Ambulatory surgical centers are expected to witness the fastest CAGR throughout the forecast period.
Impact of AI on the Carpal Tunnel Release Systems Market
AI is playing a transformative role in the Carpal Tunnel Release (CTR) Systems Market, enhancing surgical precision, patient outcomes, and overall efficiency. From preoperative diagnosis to post-surgical rehabilitation, AI-driven innovations are optimizing every stage of treatment. Below are some key ways AI is influencing the market:
1. AI-Powered Diagnostics and Early Detection
AI-based imaging and machine learning algorithms are improving the early detection of carpal tunnel syndrome (CTS). AI can analyze nerve conduction studies, electromyography (EMG) results, and wrist imaging scans to detect subtle signs of nerve compression, leading to earlier and more accurate diagnoses.
2. Robotic-Assisted Carpal Tunnel Release Surgery
AI-driven robotic surgical systems enhance precision during carpal tunnel release procedures. These systems use real-time data, motion tracking, and AI-guided navigation to assist surgeons in making smaller, more precise incisions, reducing recovery time and improving patient outcomes.
3. Personalized Treatment Planning
Machine learning algorithms analyze patient data to create personalized treatment plans. AI can assess factors such as nerve damage severity, patient health history, and expected recovery time, helping surgeons determine the most suitable treatment—whether it’s open surgery, endoscopic release, or conservative management.
4. AI in Post-Surgical Rehabilitation
AI-powered rehabilitation platforms and wearable devices track patients’ recovery progress. These systems use motion sensors and AI analytics to monitor wrist mobility, detect complications, and provide real-time feedback to patients and healthcare providers, ensuring optimal recovery.
5. Predictive Analytics for Surgical Outcomes
AI-driven predictive analytics help anticipate patient outcomes based on past surgical data. By analyzing thousands of previous carpal tunnel release cases, AI can predict potential risks, optimize surgical techniques, and improve decision-making for better long-term results.
6. AI in Minimally Invasive Techniques
Endoscopic carpal tunnel release systems benefit from AI-assisted image recognition and robotic precision. AI enhances visualization during surgery, allowing for improved accuracy, reduced complications, and faster recovery times.
7. AI-Driven Market Growth and Innovation
AI is driving innovation in the development of new CTR systems by aiding in the design of more efficient, patient-friendly surgical tools. Companies are using AI-driven simulations and modeling to improve instrument ergonomics, reduce surgical invasiveness, and enhance procedural success rates.
Growth Factors
The Carpal Tunnel Release (CTR) Systems Market is experiencing significant growth due to the rising prevalence of carpal tunnel syndrome (CTS), particularly among office workers, factory laborers, and individuals performing repetitive hand movements. The aging population and the increasing incidence of conditions like diabetes and arthritis, which contribute to CTS, are also driving the demand for CTR procedures. As more people become aware of the symptoms and risks associated with CTS, early diagnosis rates are improving, further boosting the market.
Technological advancements in minimally invasive surgery (MIS) are a major growth driver, as procedures like endoscopic carpal tunnel release (ECTR) offer faster recovery times, reduced scarring, lower infection risks, and minimal postoperative pain. Innovations such as AI-assisted robotic systems, ultrasound-guided procedures, and advanced imaging techniques are enhancing surgical precision and improving patient outcomes. These technological improvements have made CTR procedures more efficient and accessible, increasing their adoption in healthcare settings.
Rising healthcare expenditure and favorable insurance policies are also propelling market expansion. Many health insurance providers now cover CTR procedures, making them more affordable for patients. Additionally, the rapid expansion of ambulatory surgical centers (ASCs) is playing a crucial role in market growth, offering cost-effective, outpatient-based CTR procedures that provide convenience and efficiency compared to traditional hospital settings. As a result, ASCs are becoming the preferred choice for CTR surgeries, further accelerating market growth.
The increasing integration of AI and robotics in surgery is transforming the CTR market. AI-driven predictive analytics, robotic-assisted surgical systems, and automated post-surgical monitoring are enhancing efficiency, reducing errors, and improving patient recovery times. Furthermore, government initiatives promoting workplace safety, musculoskeletal health, and occupational therapy programs are contributing to the increased adoption of CTR systems. With continued advancements in minimally invasive techniques, AI-powered solutions, and expanding outpatient care facilities, the CTR systems market is poised for sustained growth in the coming years.
Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 1,74 Million |
| Market Size in 2025 | USD 980 Million |
| Market Size in 2024 | USD 920 Million |
| Market Growth Rate from 2025 to 2034 | CAGR of 6.60% |
| Dominated Region | North America |
| Fastest Growing Market | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Product Type, End user, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America and Middle East & Africa |
Market Dynamics
Market Drivers
One of the primary drivers of the market is the increasing incidence of CTS due to repetitive hand movements, prolonged computer usage, and workplace-related strain. Additionally, the rising adoption of minimally invasive techniques, which offer faster recovery and reduced post-operative pain, is fueling demand. Technological advancements, including AI-assisted surgical planning and robotic-guided procedures, are further contributing to market growth. The expansion of ambulatory surgical centers and improved insurance coverage for carpal tunnel release procedures are also key factors driving the market.
Market Opportunities
The growing preference for outpatient surgeries is creating significant opportunities for market players to develop cost-effective and efficient CTR solutions. The increasing integration of AI and robotics in surgical procedures is paving the way for enhanced precision and better patient care. Additionally, the expansion of telehealth and remote patient monitoring systems is opening new avenues for post-surgical rehabilitation and follow-up care. Companies investing in AI-powered diagnostic tools for early detection of CTS can gain a competitive edge in the market.
Market Challenges
Despite its growth, the market faces several challenges, including the high cost of advanced surgical systems, which may limit adoption, particularly in developing regions. A shortage of skilled professionals trained in minimally invasive and robotic-assisted CTR procedures is another obstacle. Furthermore, post-surgical complications and the risk of nerve damage continue to be concerns, requiring further technological improvements. Stringent regulatory requirements for new medical devices and surgical techniques also pose challenges for market expansion.
Regional Outlook
North America dominates the market due to the high prevalence of CTS, advanced healthcare infrastructure, and early adoption of robotic-assisted surgical techniques. Asia Pacific is expected to grow at the fastest rate, driven by a rising geriatric population, increasing healthcare expenditure, and growing awareness of minimally invasive procedures. Europe is also experiencing significant growth due to advancements in surgical technology and favorable government initiatives supporting musculoskeletal health.
Carpal Tunnel Release System Market Companies

- A.M. Surgical Inc.
- Arthrex, Inc.
- CONMED Corporation
- Innomed, Inc.
- Integra LifeSciences
- Medical Designs LLC
- MicroAire Surgical Instruments, LLC
- PAVmed Inc.
- S2S Surgical LLC
- Smith & Nephew plc
- Sonex Health, LLC
- Spirecut
- Stryker
- Trice Medical
Latest Announcements by Industry Leaders
- March 2025 – JUNE Medical
- Founder and Owner of JUNE Medical – Angela Spang
- Announcement – JUNE Medical has announced the upcoming launch of the HandMe Galaxy II Retractor, a device aimed at enhancing access and visibility during hand surgeries. Angela Spang, founder and owner of JUNE Medical, stated, “As an inventor, there is nothing more rewarding than creating devices that make surgery safer and easier. It is an honor to be trusted to assist surgeons in over 40 countries as they perform miracles daily.”
Recent Developments
- In December 2023, Sonex Health celebrated a significant milestone in the treatment of carpal tunnel syndrome (CTS), as they reached over 20,000 patients treated with their UltraGuideCTR device for carpal tunnel release (CTR) using real-time ultrasound guidance. This achievement follows a successful USD 40 million Series B financing round, with funds allocated to enhance access to the company’s commercially available devices designed to safely and effectively treat patients suffering from CTS and trigger finger (TF).
- In June 2022, Sonex Health announced the full U.S. commercial launch of the UltraGuideTFR, an ultrasound-guided device for trigger finger release procedures. This device allows surgeons to visualize anatomy before making an incision, resulting in smaller incisions and more minimally invasive surgery for patients undergoing trigger finger release.
Segments Covered in the Report
By Product Type
- Open Carpal Tunnel Release Systems
- Endoscopic Carpal Tunnel Release Systems
By End User
- Hospitals
- Ambulatory Surgical Centers
- Other
By Geography
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
Also Read: Site Management Organization Market
Ready for more? Dive into the full experience on our website@ https://www.precedenceresearch.com/